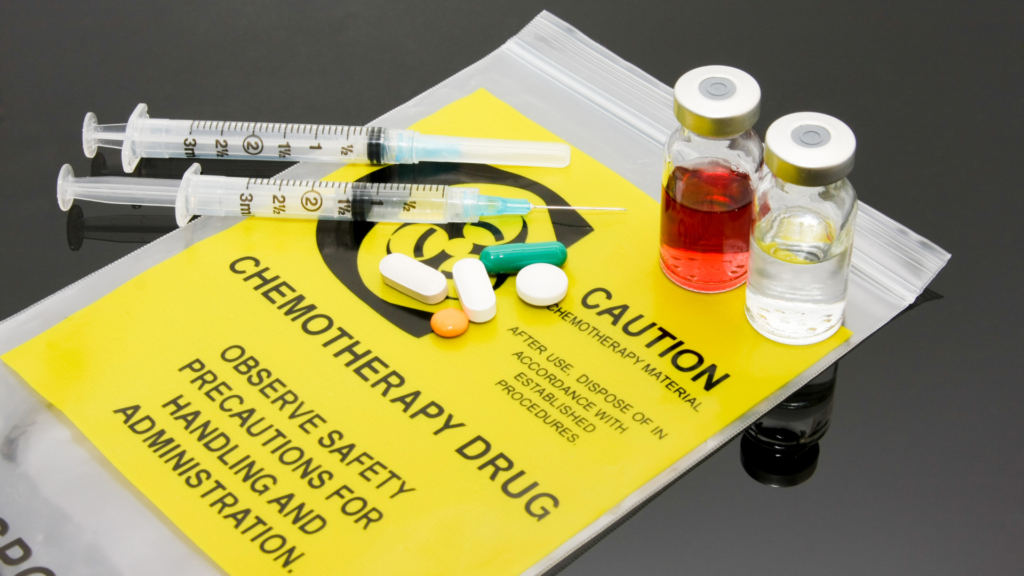
Every dental office creates pharmaceutical waste, from the buzzing multi-chair clinic to the quiet two-chair practice. This vital yet often overlooked component of dental practice operations includes expired medications, used syringes, and empty anesthetic cartridges. The improper disposal of these items creates a significant environmental and public health concern. Moreover, it can lead to substantial fines and legal complications.
Given the potential risks associated with improper pharmaceutical waste management, dental offices must have robust systems for safe and compliant handling. This guide will explore the regulatory landscape, provide practical strategies for pharmaceutical waste management, and highlight how partnering with a professional waste management company like AMS MedWaste can help dental offices navigate this complex issue.
Navigating the Regulatory Landscape
The Environmental Protection Agency (EPA) has strict guidelines for managing and disposing of pharmaceutical waste. The Resource Conservation Recovery Act (RCRA), revised in December 2018, mandates that dental offices, like hospitals and nursing homes, follow these guidelines. Notably, the RCRA prohibits flushing hazardous waste pharmaceuticals (HWPs) down toilets or drains, a practice contributing significantly to water pollution.
However, these regulations are not just guidelines – they carry the weight of the law. Non-compliance with the RCRA can result in hefty fines, as much as $37,500 per day per violation. These penalties serve as a strong deterrent against non-compliance, underlining the seriousness of these regulations.
Overall, navigating the regulatory landscape of pharmaceutical waste management is paramount for healthcare facilities. By understanding and adhering to the guidelines set out by the EPA and the RCRA, dental offices can avoid hefty fines and contribute to a healthier, safer environment for all.
Implementing Effective Pharmaceutical Waste Management
Dental offices need to establish comprehensive systems and policies for pharmaceutical waste disposal to avoid potential hazards and hefty fines. The first step is segregating waste into hazardous and non-hazardous categories, as outlined by the RCRA regulations.
Hazardous pharmaceutical waste includes RCRA-listed pharmaceutical waste, bulk chemotherapy waste, and controlled substances. RCRA-listed waste contains substances listed on RCRA’s acute or toxic waste lists as the primary active ingredient. Bulk chemotherapy waste contains more than three percent of a P or U-listed chemotherapy product. Controlled substances are drugs regulated by the Drug Enforcement Administration (DEA) due to their potential for abuse or addiction.
Non-hazardous waste includes drugs that do not fall under the EPA RCRA waste ID for characteristic, acute, or toxic listing; products deemed non-hazardous in the work environment by the Occupational Safety and Health Administration (OSHA); and products classified as non-carcinogenic by the US Department of Health.
Partnering with AMS MedWaste for Comprehensive Pharmaceutical Waste Management
At AMS MedWaste, we understand the complexities of pharmaceutical waste management. We offer dental offices a comprehensive solution to ensure compliance with EPA regulations and safe disposal of pharmaceutical waste. Our services, available to clients in Illinois, Southern Wisconsin, and Northern Indiana, include waste segregation, pickup and transportation, and disposal.
Our team takes care of the entire waste management process, allowing dental offices to focus on their primary mission – providing exceptional dental care for their patients. We ensure safe, efficient, and compliant pharmaceutical waste disposal, helping dental offices avoid potential health hazards, hefty fines, and legal complications.
If you’re a dental office looking to improve your pharmaceutical waste management practices or have questions about our services, we invite you to contact us today. Let’s work together to create a safer, cleaner, and environmentally friendly dental practice.